Methanol (MeOH)
With a global demand of 60 million tonnes, methanol is one of the most commonly produced organic chemicals (as of 2012) and is used as a raw material in the production of basic chemicals such as formaldehyde and acetic acid for the chemical industry. In addition to its material use, methanol and its daughter products can also be used as an energy source. Methanol produces fuel using “Methanol to Gasoline” technology. It supplies fuel cells with hydrogen. Methanol is also required in the synthesis of biodiesel.
| Most common applications: |
Formaldehyde production Acetic acid production Solvents Fuel Waste water treatment |
| Other names: |
Methyl alcohol Carbinol
|
| Molecular formular: |
CH4O |
| CAS number: |
67-56-1 |
This information reflects the current state of our experience, however, this does not mean it guarantees certain properties of the product or its suitability for any specific purpose. This information does not absolve the user from the obligation of conducting the customary receiving inspection on acceptance of our products.
| Brief description: | colorless liquid |
| Molecular weight: |
32.04 g•mol−1
|
| Physical state: | liquid |
| Density: |
0.79 g / cm³ |
| Melting point: |
−98° C |
| Boiling point: |
65° C |
| Vapour pressure: |
129 hPa (20° C) |
| pKa value: |
16 |
| Solubility: |
miscible with water, ethanol and diethyl ether |
| Refractive index: |
1,3288 (20° C) |
This information reflects the current state of our experience, however, this does not mean it guarantees certain properties of the product or its suitability for any specific purpose. This information does not absolve the user from the obligation of conducting the customary receiving inspection on acceptance of our products.
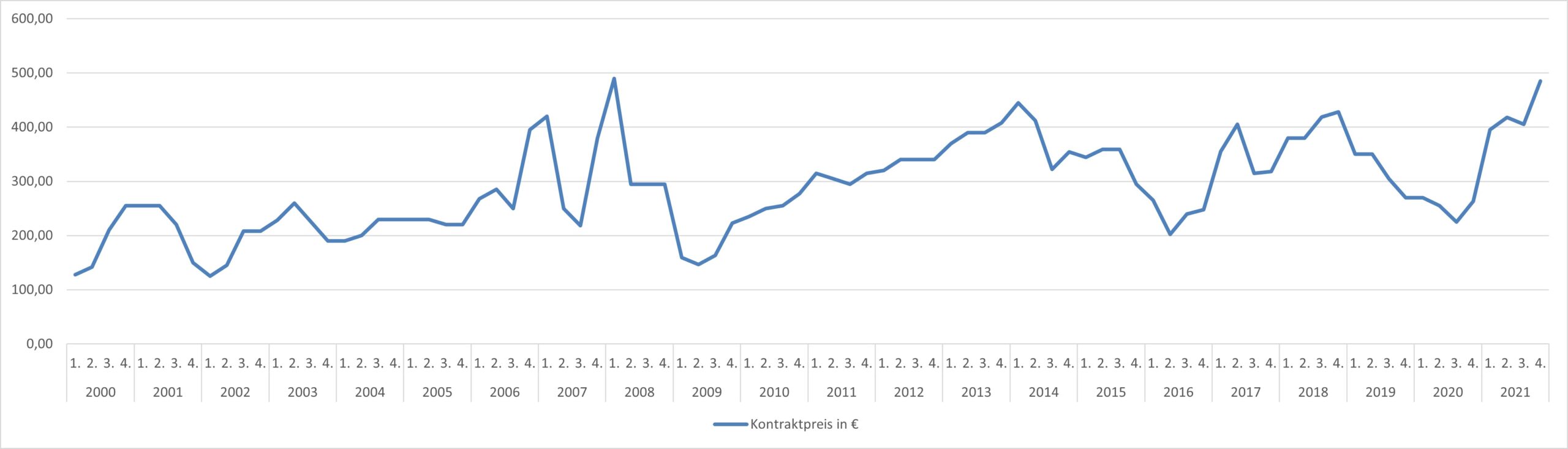
Historical Methanol Pricing